MCW Genomic Sciences & Precision Medicine Center (GSPMC)
Under the umbrella of the Precision Medicine Initiative (PMI) at the Medical College of Wisconsin (MCW), the Genomic Sciences and Precision Medicine Center (GSPMC) offers campus clinicians, and researchers a consolidated one stop shop for clinical testing and research investigation.
With clinical-grade services in genomics, transcriptomics, epigenomics, and molecular pathology, all performed within a CAP/CLIA accredited laboratory, GSPMC not only fulfills your patient testing needs but also partners with you to design needed novel clinical assays, customize your research projects, and analyze and report your data.
GSPMC works with physicians, genetic counselors, allied health professionals, and researchers across interdisciplinary teams in the medical and academic communities, nationally and internationally, to advance the science of Precision Medicine, paving the way for the emerging world of data-driven healthcare.
 COVID-19 Assays
COVID-19 Assays
Amidst the recent escalation of the Coronavirus Pandemic, significant research and clinical testing needs have emerged. In an effort to continue its steadfast support on and beyond the Medical College of Wisconsin’s (MCW) campus, the GSPMC has expanded its technological portfolio and offering of assays to facilitate COVID-19-related investigations. Along with these new services, the Center is committed to delivering physicians and researchers the latest technology, shortest feasible turnaround time, and quality customer service.
COVID-19
- Coronavirus disease 19 (COVID-19), a respiratory illness is caused by Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
- SARS-CoV-2 is positive–sense single-stranded RNA virus, that enters human cells by binding to the angiotensin converting enzyme 2 (ACE2) receptor.
- Common symptoms of infection include fever, tiredness and dry cough.
Assays
- Detection of SARS-CoV-2 RNA in nasopharyngeal (NP) swabs [Research Use Only]
- Genotyping of ACE polymorphisms (I/D) in whole blood and NP swabs
- Evaluation of ACE2 receptor expression in whole blood
- Whole genome sequencing of SARS-CoV-2 for strain typing and surveillance in NP swabs [Research Use Only]
Specimen Requirements
| Specimen Type | Shipping Temperature | Container | Special Requirements |
|---|---|---|---|
| Nasopharyngeal Swabs | -20C or -80C | Collection should use a flocked or spun polyester swab stored in Universal Transport Medium (UTM) | Minimum of 1mL of UTM received within 72 hours of collection. |
| Whole Blood | Ambient | Purple top EDTA tube. Insulated shipping container. | 2-4mL of whole blood received within 5 days of draw. |
| gDNA* | Ambient | Screw cap tube. | 10μg of purified gDNA, > 75ng/μL, 260/280 purity ratio of 1.75-2.0. |
| RNA | -20C or -80C | Screw cap tube. | 500ng of purified RNA, >25ng/uL. |
COVID-19 Assays

SARS-CoV-2 virus detection (qRT-PCR)
- A quantitative reverse transcription PCR (qRT-PCR) assay to evaluate for the presence of the SARS-CoV-2 virus, specifically nucleocapsid (N) gene regions N1 and N2.
- The assay is intended for the qualitative detection of SARS-CoV-2 RNA in nasopharyngeal swabs; research specimens only (https://www.cdc.gov/coronavirus/2019-nCoV/lab/index.html)
- Turn-around time (from sample receipt to result) – 6hrs
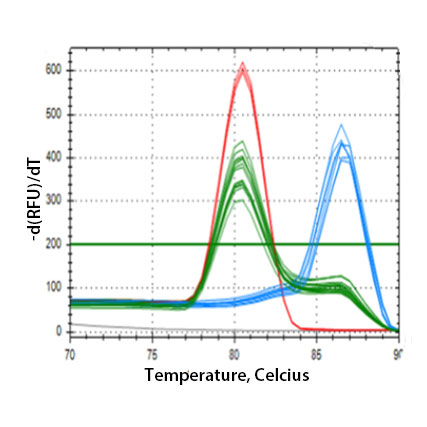
ACE Polymorphism Genotyping (qPCR)
- A quantitative PCR to identify the genotype of a functional polymorphism present in intron 16 of the ACE gene, in the form of insertion (I allele) and/or deletion (D allele) of 289 bp Alu repeat sequence (rs4646994; rs1799752; rs4340 and rs13447447 associated with the same polymorphism) (PMID:24498151) .
- The I/D polymorphism may affect the expression of ACE gene and/or the function of angiotensin I converting enzyme (PMID:17504229). The presence of the D allele is associated with reduced expression of ACE2 (PMID:10969042)
- Turn-around time (from sample receipt to results) – 6 hrs
Assays Under Development
ACE2 Receptor Expression (qRT-PCR)
- A quantitative reverse transcription PCR (qRT-PCR) assay to evaluate the level of ACE2 receptor expression on peripheral blood monocytes.
- The angiotensin converting enzyme 2 (ACE2) is expressed on the surface of lung alveolar epithelial cells and enterocytes of the small intestine as well as in the kidney heart and blood vessels (PMID:1514137) and serves as the host receptor for SARS-CoV-2 (PMID:32142651).
- Estimated date of availability of assay: May 15, 2020

Whole Genome Sequencing of SARS-CoV-2 (NGS)
- A 343-amplicon, highly-multiplexed PCR-based assay for NGS analysis of the entire SARS-CoV-2 genome.
- High sensitivity (can detect down to one copy), and it provides >97% coverage for 107 known coronavirus strains.
- Full sequencing would enable surveillance and monitoring of viral evolution.
- Estimated date of availability of assay: May 15, 2020
Request Services
Please sign in with your CTSI credentials to access this form. If you haven't signed up yet, please join CTSI.
NIH Funding Acknowledgment: Important Reminder – Please acknowledge the NIH when publishing papers, patents, projects, and presentations resulting from the use of CTSI resources by including the NIH Funding Acknowledgement.





