17 Apr Funding Opportunity: GPC 2017 Inter-Institutional Pilot Program RFP
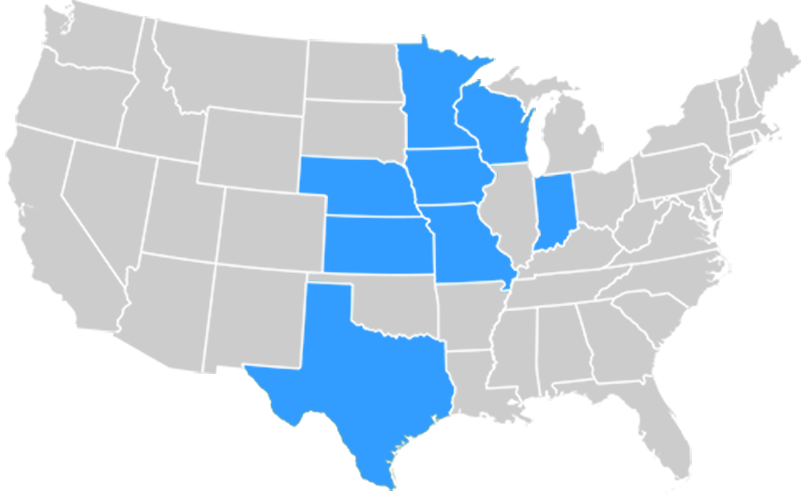
The Greater Plains Collaborative (GPC) is a network of twelve leading medical centers in eight states committed to a shared vision of improving healthcare delivery through ongoing learning, adoption of evidence-based practices, and active research dissemination.
Partners by state
- Kansas – University of Kansas Medical Center (KUMC)
- Missouri – Children’s Mercy Hospital; University of Missouri
- Iowa – University of Iowa Healthcare
- Wisconsin – University of Wisconsin-Madison; Medical College of Wisconsin; Marshfield Clinic
- Minnesota – University of Minnesota Medical Center
- Nebraska – University of Nebraska Medical Center
- Indiana – Indiana University
- Texas – University of Texas Health Sciences Center at San Antonio; University of Texas Southwestern Medical Center
With support from our Clinical Translational Science Institutes’ leadership (NIH CTSA and CTR programs), the GPC network is seeking to broaden awareness and utilization by investigators at its partner institutions to capitalize on our heightened patient engagement and unique data resources.
Key Dates
- Announcement Date: April 10, 2017
- Informational Webinars for Potential Applicants: May 3, 2017 @ 3 pm – 4 pm CDT; May 10 @ 9 am – 10 am CDT
- Letter of Intent Due Date: May 31, 2017
- Application Due Date: July 21, 2017
- Announcement of Finalists: August 31, 2017
- Awardee Participation at GPC Learning Exchange Conference: October 16-17, 2017 (Location: Kansas City, Mo; In-person attendance requested, travel funds provided)
- Project Start Date: September 1, 2017
- Project End Date: August 31, 2018
Program
Program
Summary: The purpose of the pilot program is to provide modest support that will allow an investigator the opportunity to develop sufficient preliminary data utilizing the GPC’s data infrastructure as a basis for a larger application for independent research support for the investigator and the network. It is not intended to support or supplement an ongoing research project of an established investigator. A proposed pilot and feasibility study should present a testable hypothesis and clearly delineate 1) the question being asked; 2) the procedures to be followed; 3) how the data will be analyzed; 4) the linkage to the national FOA/RFA for which the pilot is strengthening competitiveness; and 5) the intended submission date for responding to the FOA/RFA. An emphasis is being placed on multi-institutional collaboration across the various GPC institutions that strengthen relationships and patient-centered collaborations amongst investigators and patients across the GPC institutions.
Award categories (with estimated approximate value):
- <$5,000 award: Pre-research queries, including feasibility queries, engagement, and patient/investigator match-making. May utilize the GPC Reusable Study Environment (GROUSE) environment of integrated claims, EHR, and registry data for breast cancer, ALS, and obesity research topics.
- $10,000 award: To support proposed observational studies, including cross-sectional, epidemiological, health services, and comparative effectiveness research. Applying with CMS for reuse of GROUSE for topics outside our current IRB protocol.
- $20,000 award: To support proposed interventional studies, including clinical trials and pragmatic randomized trials by piloting patient reported outcome measures and/or surveys at multiple sites.
GPC institutions have agreed to provide in-kind support for informatics resources and, in some locations, matching funding for their institutional investigators.
The number of awards will be dependent upon the quality of the proposals received and the final total of committed matching funds. It is anticipated that, at a minimum, two awards in each category will be made.
Applicants are strongly encouraged to make initial contact with their appropriate institutional Greater Plains Collaborative Site PI to discuss their project prior to submitting a LOI/application.
Eligibility and Allowable Costs
Eligibility
- New investigators without significant peer-reviewed
- Established investigators with no previous work utilizing the GPC/PCORnet infrastructure.
- Established investigators who have utilized their local informatics resources and are interested in expanding their projects to include data/collaborators from other GPC institutions.
Each pilot proposal should state clearly the justification for eligibility of the investigator under one of the above three criteria. An investigator may submit one application as principal investigator but is encouraged to participate in multiple proposals as a co-investigator with other GPC institutions provided that each application is scientifically distinct.
Allowable Costs
- The maximum award is $20,000 in direct No F&A costs will be awarded.
- Salary support and fringe benefits for research assistants/associates, postdoctoral fellows, or students
- Research materials and supplies
Awardees have discretion in the budgeting of funds to meet their research needs within the general guidelines of the budget and the terms of the proposal; however, funds may not be transferred to another project.
Review and Award
Review and Award
Research proposals will be initially reviewed by a committee comprised of GPC representatives and colleagues at affiliated CTSAs/institutions. The reviewers will evaluate proposals based on the following criteria:
- Scientific merit/quality of research plan (20 points)
- Validity and feasibility of the approach
- Significance and relevance of the research
- Probability of extramural funding, if successful (20 points)
- Innovativeness of the project
- Expertise of the investigator
- Utilization of the GPC’s infrastructure to increase research generalizability (20 points)
- Patient engagement (20 points)
- Value added by a multi-institutional approach (20 points)
Presentation of awarded projects is requested at the GPC’s 2017 Learning Exchange Conference (October 16-17, 2017). This will provide the opportunity to interact with patients and network/informatics leaders to further strengthen all proposals.
Travel to Kansas City to attend the GPC Learning Exchange Conference will be reimbursed by the Greater Plains Collaborative and thus isn’t required to be a part of the proposed pilot grant budget.
REPORTING
Awardees will submit a brief report, including research progress, budget expenditures, publications, and new or proposed external support by September 30, 2018.
ACKNOWLEDGEMENT OF SUPPORT
Any publication arising from work supported by the grant should acknowledge the PCORI Greater Plains Collaborative Clinical Data Research Network CDRN-1306-04631.
How to Apply
How to Apply
General instructions
Applications must be in Arial font, size 11 point or larger, with margins of at least 0.5 inch.
- Letter of Intent (LOI)
A non-binding letter of intent is required by May 31st 2017. The LOI should briefly (400 words or less) but adequately describe the proposed research project and requests for collaborating partners. Please submit the LOI as a .pdf file via e-mail to gpcnetwork@kumc.edu. - Application Cover Page
The cover page form must be filled out, scanned, and attached to the front of the application.
81 KBGPC 2017 Pilot Program Application Cover Sheet - Summary (one-half page or less)
Describe the primary objective of the proposed research, the key components of the research design, and its significance in general terms. - Budget
Provide a budget explanation on a separate page in collaboration with your GPC site PI. Clarify the role of individuals for whom salary support is requested. Equipment, if included, must be well justified. No specific format is required so long as the budget and explanation contain the relevant information. - Other Support
On a separate page, list all other sources of active or pending internal and external support for the PI and other key personnel. For each, give the title, sponsor, role of the PI on the project, funding period, annual direct costs, percent effort by the PI, and the goals of the project. Describe the relationship of each to the current proposal. In cases in which existing funds appear to be similar to the proposed project, take special care to explain the differences. - Biographical Sketch
Provide a biographical sketch for the PI in the latest NIH format. - Research Strategy (Up to 5 single-spaced pages for Items A.-D. Proposals may not
exceed that length)- A. Specific Aims (one-half page recommended)
List the broad, long-term objectives and what the specific research proposed in this application is intended to accomplish. - B. Significance
State the importance and relevance of the research project. - C. Innovation
Explain how the application challenges current research or clinical practice paradigms. Describe any novel concepts, approaches, instrumentation or intervention(s) to be developed or used. - D. Approach
Describe the research activities to be carried out under the award. State your research design, and explain how it will allow you to address the question(s) and hypotheses of the project. Explain potential pitfalls you might encounter and how you plan to overcome them. Describe how GPC data infrastructure and multi-institutional collaboration strengthens your research’s competitiveness. Include your patient engagement strategy regarding design, implementation, and/or dissemination of the research. Preliminary studies can be included but are not required.
- A. Specific Aims (one-half page recommended)
- Appendix
Include surveys, other measurement instruments and similar items. The Appendix may not be used to provide additional data or information that should be part of the 5 page proposal. - Letter(s) of Collaboration
While not required, letters from the other GPC site co-investigators engaged are welcome. - Letter(s) from your CTSI and GPC site
This should indicate support for the PI to do the research. Indicate any commitment of matching or contributed funds to support the research project by your campus. - Status of IRB or IACUC Submission
If applicable, please provide updated documentation on where the proposal is in the approval process. If approval has been granted, include confirming documentation. If approval is not complete, indicate whether or not it is currently in process and if so submit documentation and a draft of the IRB application. If not yet approved, indicate if there are any specific foreseeable challenges in obtaining approval. - Bibliography (not to exceed 2 pages)
Application Submission
All materials must be submitted electronically as a single document in Adobe Acrobat (.pdf) format to gpcnetwork@kumc.edu.
Pre-submission inquiries should be directed to your local GPC Site PI.
For logistical information regarding the informational webinars or other program questions, please contact Steve Fennel, Greater Plains Collaborative Clinical Data Research Network/University of Kansas Medical Center, sfennel@kumc.edu.
Acknowledgements: The Greater Plains Collaborative acknowledges both Frontiers: The Heartland Institute for Clinical and Translational Research and the Kansas PKD Center at the University of Kansas Medical Center for the pilot program framework.